Privacy-safe synthetic data that accelerates evidence generation.
Aindo enables pharmaceutical companies, CROs, and research organizations to generate high-quality Real-World Evidence while maintaining full compliance with data protection and regulatory requirements.
Whether you're developing new therapies, validating treatment effectiveness, or accelerating time to market, we help you work faster, safer, and with greater confidence.
With Aindo's AI evidence infrastructure, you can:
Built for Europe's advanced healthcare regulations.
Designed for the new European health data framework and certified for data processing in healthcare.

Europrivacy
Art 42 GDPR
ISO 9001
Quality
ISO 27001
Information security

Gender Equality
UNI PDR 125:2022
NIST
Cybersecurity & privacy
Built for teams driving pharma evidence
Designed for RWE, clinical development, HEOR, market access, and medical affairs teams.
RWE & HEOR teams
Run comparative analyses, outcomes research, and population-level studies without access delays.
Pharma & CRO clinical teams
Assess feasibility, reduce recruitment risk, and accelerate study design decisions.
Medical affairs & safety
Explore post-market signals, safety trends, and subgroup insights while remaining compliant.
From raw data to evidence-ready cohorts
Talk to our experts
Ingest and harmonize healthcare data
Clinical data are ingested and mapped to a standardized structure.
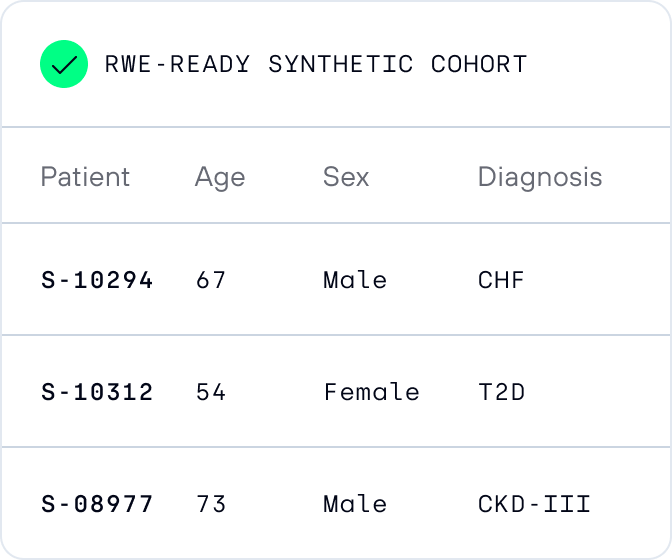
Generate and validate synthetic cohorts
Synthetic data are generated and validated for utility, bias, and privacy.
Deliver compliant, analysis-ready datasets
Teams receive cohorts ready for analysis, sharing, and iteration.




