External Control Arms
Accelerate clinical trials with validated synthetic control arms
Aindo enables the creation of robust synthetic cohorts to support faster, more efficient, and more ethical clinical studies.
Talk to our experts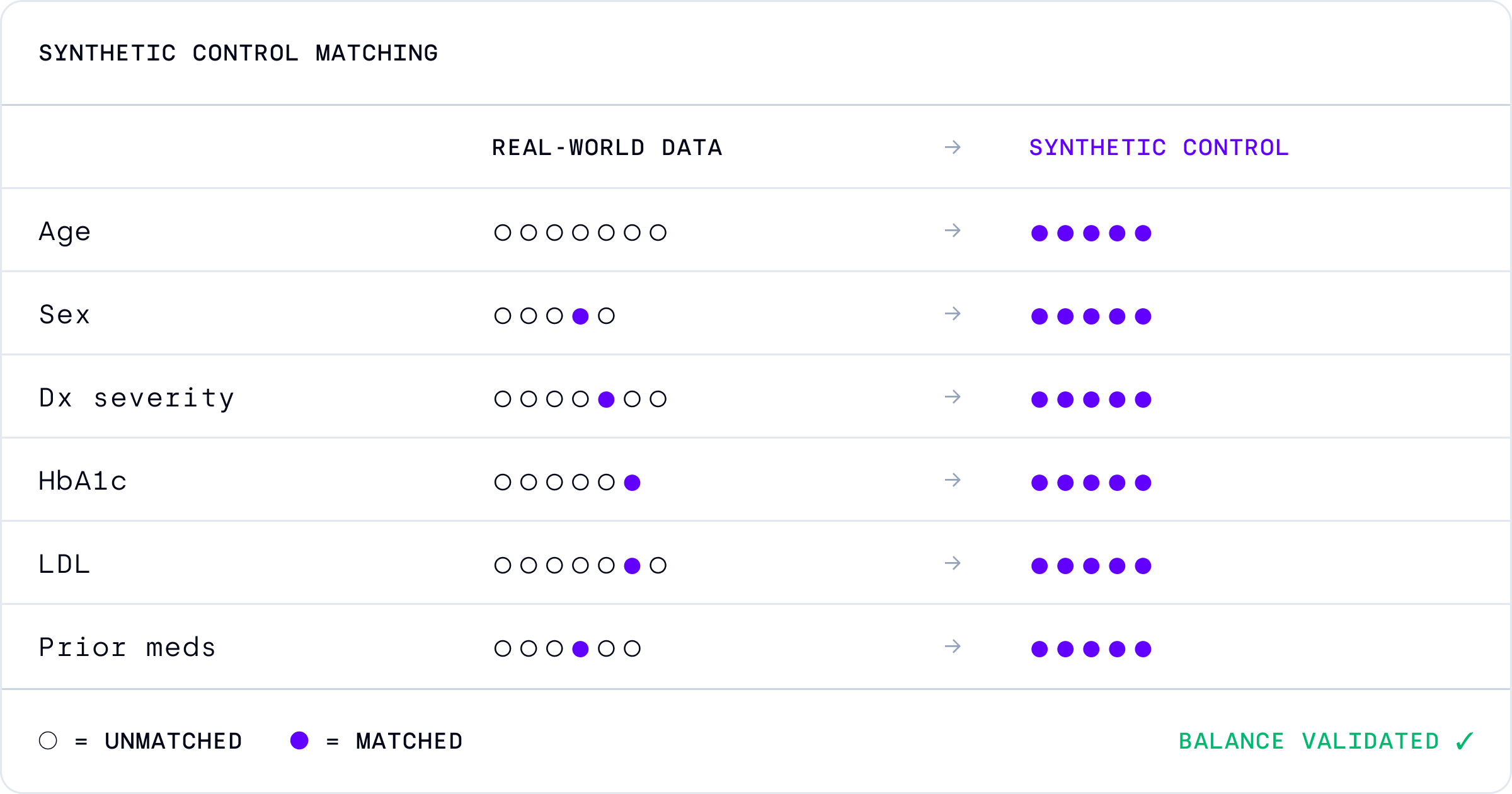
Aindo helps pharma companies, CROs, and research organizations integrate external control arms while maintaining strong methodology, privacy, and regulatory compliance.
Accelerates clinical studies Reduce cost and recruitment burden Improve operational efficiency
With Aindo's AI evidence infrastructure, you can:
Built for Europe's advanced healthcare regulations.
Designed for modern trials and aligned with the highest European standards for health data and clinical research.

Europrivacy
Art 42 GDPR
ISO 9001
Quality
ISO 27001
Information security

Gender Equality
UNI PDR 125:2022
NIST
Cybersecurity & privacy
Use synthetic control arms to
Support single-arm trials and rare disease studies
Strengthen comparative effectiveness analyses
Reduce placebo exposure
Improve statistical power and cohort representativeness
Enable faster regulatory submissions







