From raw data to evidence-ready cohorts
Our AI evidence infrastructure allows you to safely access, analyze, and share health data.
Talk to our team
Aindo enables pharmaceutical companies, CROs, and research organizations to generate high-quality Real-World Evidence while maintaining full compliance with data protection and regulatory requirements.
With Aindo's AI evidence infrastructure, you can:
Built for Europe's most advanced healthcare regulations
Designed for the new European health data framework and certified for data processing in healthcare.

Europrivacy
Art 42 GDPR
ISO 9001
Quality
ISO 27001
Information security

Gender Equality
UNI PDR 125:2022
NIST
Cybersecurity & privacy
Where synthetic data amplifies real-world evidence impact
Regulatory submissions
Support regulatory submissions and label expansion studies with compliant synthetic evidence.
Post-market surveillance
Enable large-scale post-marketing surveillance and pharmacovigilance analytics at scale.
Comparative effectiveness
Conduct comparative effectiveness and outcomes research across representative populations.
Health economics & market access
Strengthen HEOR, pricing, and market access models with scalable synthetic datasets.
Feasibility & protocols
Accelerate feasibility studies and optimize protocols before and during clinical trials.
Data collaboration
Enable data sharing across consortia and external partners without exposing patient data.
Explore how synthetic data supports high-quality RWE
Read our white paper on synthetic data in clinical research.
Explore our white paper
Ingest and harmonize healthcare data
Clinical data are ingested and mapped to a standardized structure.
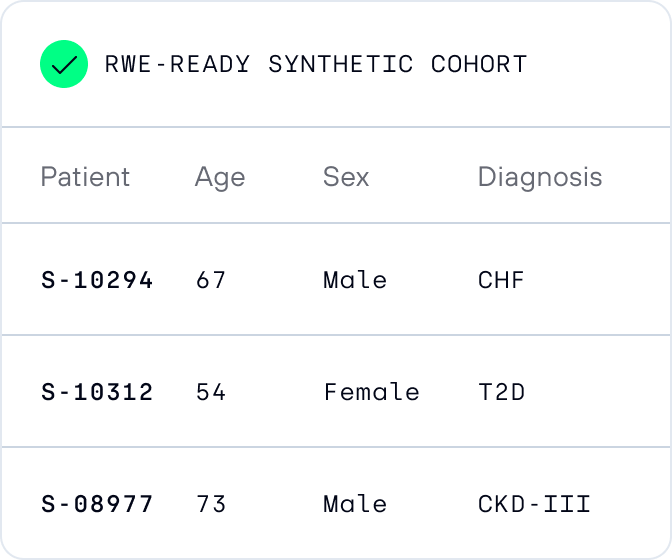
Generate and validate synthetic cohorts
Synthetic data are generated and validated for utility, bias, and privacy.
Deliver compliant, analysis-ready datasets
Teams receive cohorts ready for analysis, sharing, and iteration.





